A tuberculosis test that spots contagious cases—with limits

A single translucent blood vial resting on the edge of an expansive, sterile steel lab bench at UC Davis, tiny in comparison to the massive, shadowy📷 Photo by Tech&Space
- ★Blood test distinguishes active TB from latent infection
- ★Early-stage research, not yet clinically validated
- ★Potential to curb transmission—but sample size unknown
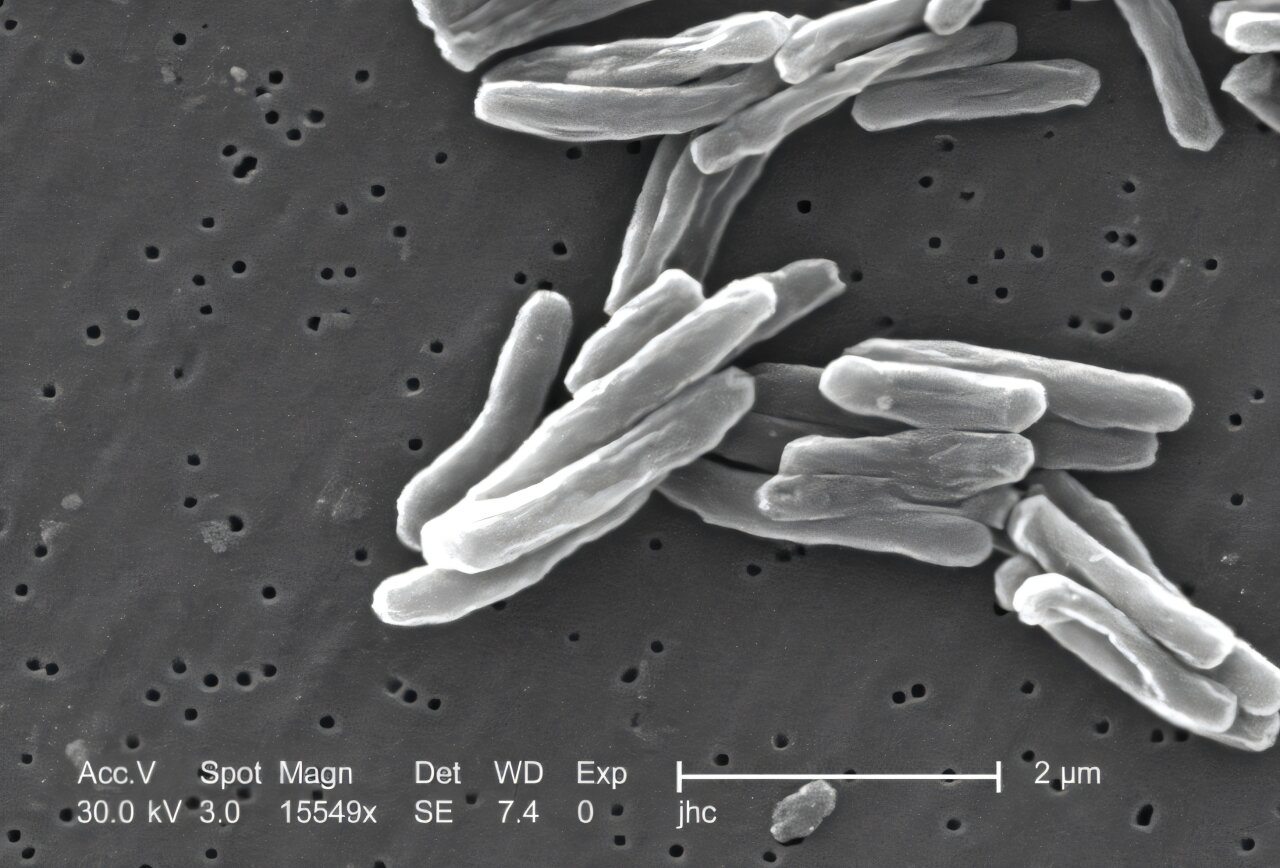
A team at UC Davis has developed a blood test that does something no current diagnostic reliably does: detect active, infectious tuberculosis (TB) while ignoring latent cases. The distinction matters. Latent TB affects a quarter of the world’s population but isn’t contagious; active TB kills 1.3 million people annually and spreads through airborne droplets. Current tests—like sputum cultures or skin prick reactions—either miss early infections or can’t differentiate between dormant and transmissible disease.
The new assay, detailed in a preprint study (not yet peer-reviewed), targets a specific immune response linked to Mycobacterium tuberculosis when it’s actively replicating. According to the researchers, this could slash diagnosis times from weeks to days. But the claim rests on a critical, unanswered question: How large was the study? The press release omits sample size, methodology details, and whether the test’s accuracy holds across diverse populations—gaps that temper the optimism.
For now, the test remains confined to lab validation. Regulatory approval would require far larger trials, including comparisons against gold-standard diagnostics like GeneXpert, which the WHO already endorses for high-burden regions. The UC Davis team’s work is a step forward—but one measured in millimeters, not miles.
A tuberculosis test that spots contagious cases—with limits📷 Photo by Tech&Space
The evidence is real, but the study’s scope is narrow
The clinical relevance today? Zero. This is research-stage technology, years away from real-world use. Even if validated, the test’s utility hinges on cost and scalability—factors the study doesn’t address. The WHO’s 2023 TB report underscores that 40% of active cases still go undetected; a faster diagnostic could help, but only if it’s affordable in low-resource settings where TB thrives.
What we don’t know outweighs what we do. The test’s sensitivity and specificity rates aren’t public. Nor is it clear whether it detects drug-resistant strains, which account for 10% of global cases. The team also hasn’t disclosed if the assay works in HIV-positive patients, who comprise 8% of TB deaths. These aren’t minor footnotes—they’re the difference between a lab curiosity and a tool that saves lives.
The real signal here isn’t the test itself, but the gap it exposes: TB diagnostics have stagnated for decades. A blood test for active disease is a long-sought goal, but this iteration is a prototype, not a panacea. The bottleneck isn’t scientific ingenuity—it’s the grind of translation: turning a ‘promising’ lab result into something a clinic in Mumbai or Lagos can actually use.