Genomic Mapping Identifies High-Risk E. coli for Vaccines
Genomic Mapping Identifies High-Risk E. coli for Vaccines📷 Published: Mar 26, 2026 at 15:08 UTC
- ★Capsule shields E. coli from immune detection
- ★Genomic mapping targets high-risk strain types
- ★Research-stage only, no patient impact yet
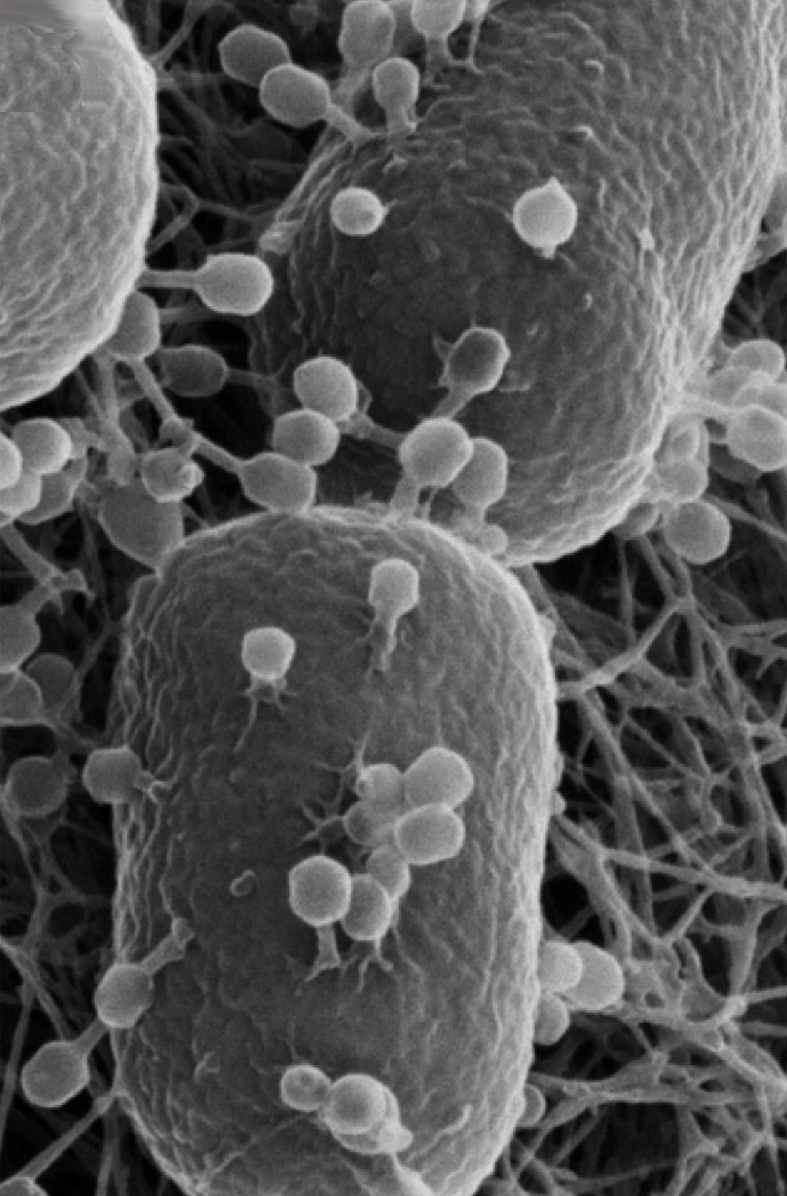
Not all E. coli strains are created equal. Researchers have now used genomic mapping to identify which capsule types make certain strains more likely to become invasive—a finding that could eventually guide vaccine development. The capsule, a sugar-rich outer layer surrounding the bacterium, acts as a shield against immune detection and some therapies, making it a critical factor in determining whether an infection becomes dangerous, according to GEN - Genetic Engineering and Biotechnology News.
The study analyzed the genomic signatures of various E. coli capsule types. Identifying these high-risk capsule variants provides a roadmap for targeting the most threatening strains. Early signals suggest this approach could streamline vaccine development by focusing efforts on the capsule types most associated with invasive disease, as detailed in recent microbiology research. The findings represent a systematic attempt to classify bacterial risk based on genetic markers rather than trial and error.

Evidence level: research stage only📷 Published: Mar 26, 2026 at 15:08 UTC
Evidence level: research stage only
What the evidence actually shows is more modest than headlines might suggest. This is genomic research—laboratory work that maps bacterial structures—not a clinical trial testing a vaccine in patients. The methodology limits matter here: the findings identify correlations between capsule types and invasiveness, but they don't prove causation or demonstrate that targeting these capsules will work in humans, a distinction often lost in science coverage.
For patients today, this changes nothing. No vaccine exists yet, and the research remains firmly in preclinical stages. What we know is that certain capsule types correlate with higher risk. What we don't know is whether vaccines targeting these capsules will prove safe, effective, or feasible to manufacture at scale. Regulatory approval, if it ever comes, would be years away following FDA guidance for biologics.